- SEIKA Digital Image Corporation.
- ProductList
- Laser-induced fluorescence method: LIF system
Laser-induced fluorescence method: LIF system

LIF (Laser Induced Fluorescence)
LIF (Laser Induced Fluorescence) is a technique to observe fluorescence from molecules by exciting specific molecules contained in the measurement target with a single wavelength light source such as a laser. Fluorescence emission is different from the wavelength of the excitation light, so it can be observed without being affected by irregular reflection of the excitation light. Measurement of gas molecular species during combustion (PLIF), micro-LIF for measuring gas concentration distribution, temperature distribution, diffusion and mixing, reaction, pH, etc. in a minute area, multi-molecular species, simultaneous measurement of temperature and velocity, etc. It is a method that can realize film thickness measurement of oil film such as multi-PLIF, bearings and oil seals.
Variation of LIF
Combustion measurement by LIF (PLIF)
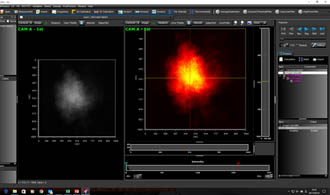
Combustion measurement by LIF makes it possible to measure gas molecular species such as acetone, OH, CH, and NO, and their instantaneous distributions. Among LIFs, planar LIF measurement using images in particular is called PLIF (image laser induced fluorescence, planar laser induced fluorescence). PLIF is a LIF system that uses ultra-sensitive cameras such as ICCD, and is a very effective method for combustion measurement and analysis.
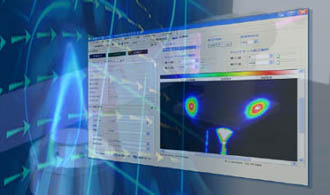 Combustion measurement by LIF : PLIF
Combustion measurement by LIF : PLIFOil film thickness measurement
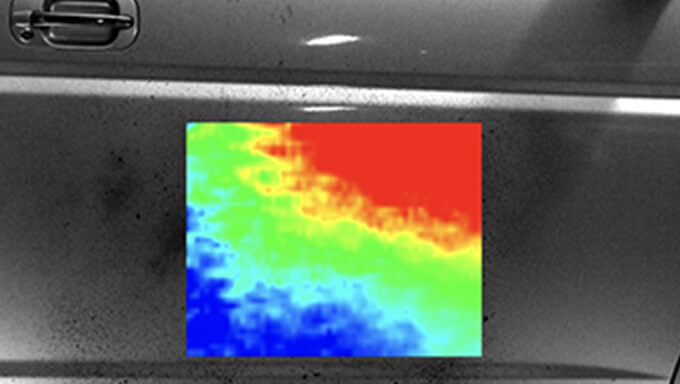
It is possible to measure the film thickness distribution of oil films such as bearings and oil seals, and visualize the spraying and adhesion conditions. Non-contact and non-destructive measurement is possible because measurement is performed by simply adding a very small amount of fluorescent dye to the measurement target. In terms of film thickness distribution, the thinnest case can be quantitatively measured to the submicron level, so it is possible to handle various films.
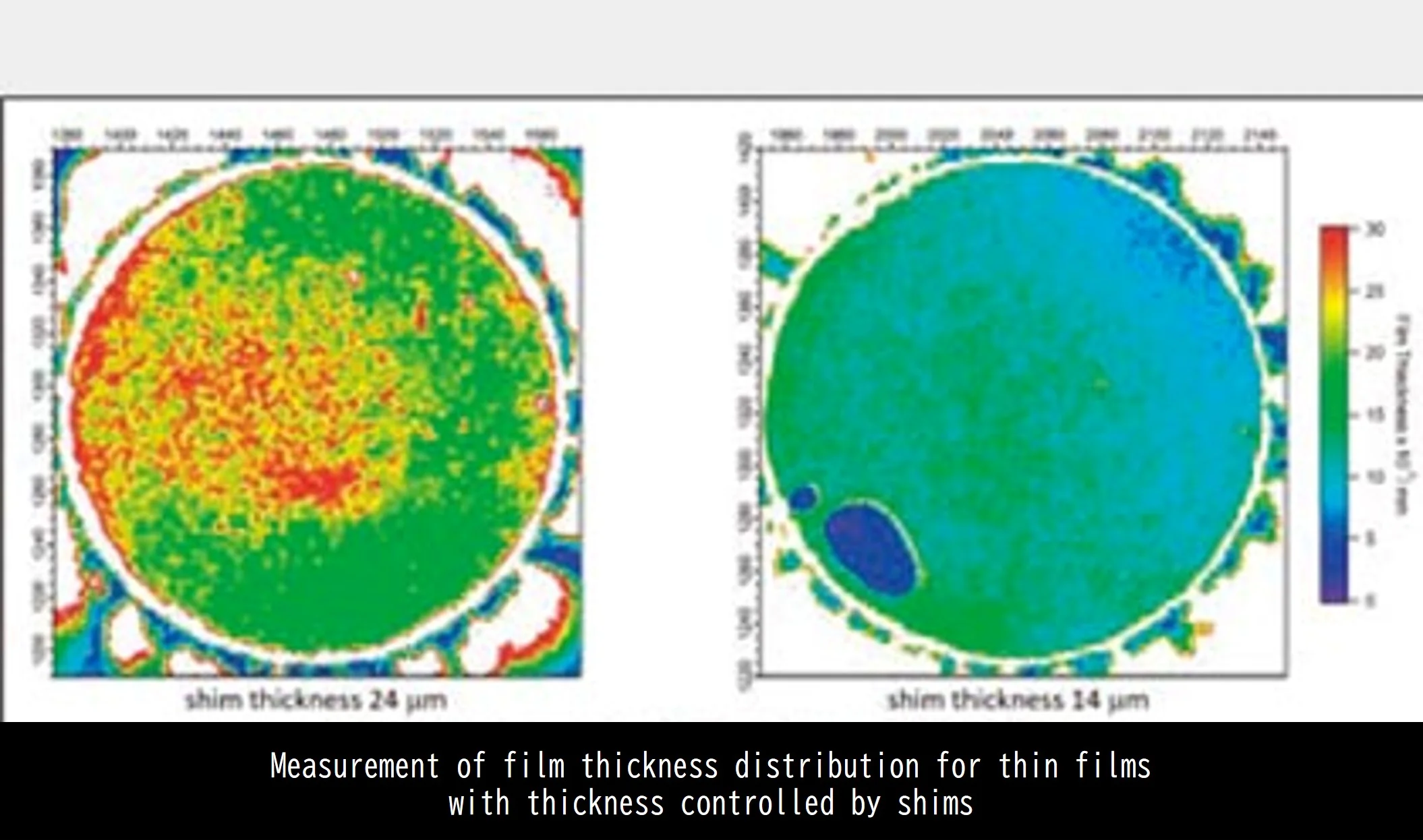 Film thickness measurement by LIF
Film thickness measurement by LIFMicro LIF
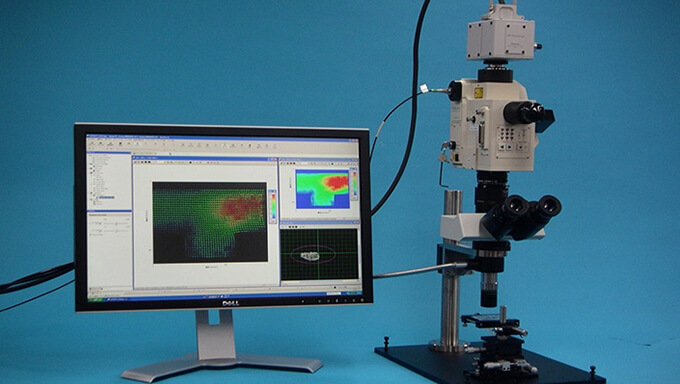
In the case of micro LIF, it is possible to measure concentration distribution, temperature distribution, diffusion, mixing, reaction, pH, etc.
The dedicated micro-optics system can co-axially illuminate a high-power UV pulse laser, which is not possible with general microscopes.

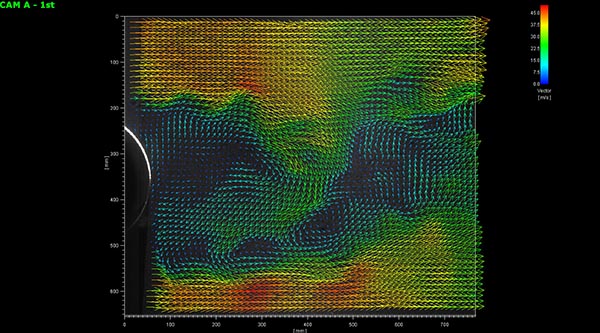
Multi PLIF
Multi-molecular species, temperature, speed, etc. can be measured simultaneously. For example, acetone exhibits an absorption peak at 280nm and fluorescence around 435nm. OH radicals are induced at 283 nm and emit fluorescence instantaneously at around 315 nm, so simultaneous measurement of these is possible.

Various methods of measuring gas molecular species
Various types exist to measure gas molecular species in flames. Point measurement, line measurement, and area measurement (2D), and also depending on the application. The table shows an example of each measurement method and measurable gas molecular species.
横スクロールでご覧いただけます。
| Measurement method | Measurement system | Gas molecular species |
|---|---|---|
| point | Emission spectrum | OH, CH, C2 |
| CARS(CoherentAnti-StokesRamanScattering) | N2, O2 | |
| line | Raman scattering method | OH, NO, NO2, C2, CH...etc |
| TDLAS(Tunable diode laser absorption spectroscopy) | CH4, CO, CO2, O2, HCl, NH3, HC...etc | |
| area |
PLIF | OH, C, NO, O2, CO See example specifications for each gas. (Japanese only) |
Here, we will introduce LIF, which is an area measurement. Combustion measurement by LIF can measure gas molecular species such as acetone, OH, CH, and NO, and their instantaneous distribution. Planar LIF measurement using images is called PLIF (Planer Laser Induced Fluorescence). PLIF is a LIF system that uses ultra-sensitive cameras such as ICCD, and is a very effective method for combustion measurement and analysis.
Features of the LIF system software "Koncerto-LIF"
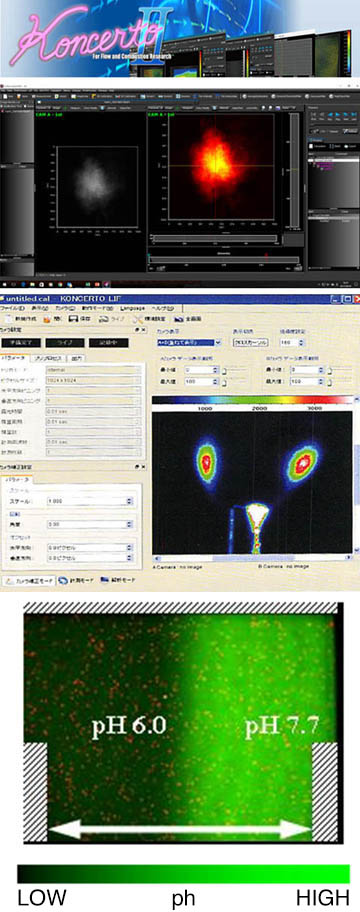
Koncerto-LIF (control and analysis software developed by Seika Digital Image) provides the functionality needed to control ultra-sensitive cameras such as ICCD and EMCCD cameras and display and analyze high-bit images. Integrated imaging measurement software.
- Camera control function
Compatible with various makes and models. Simultaneous control of multiple cameras and image display are possible. - Camera alignment function
This is an adjustment function to equalize the angle of view of the two cameras. A calibration target is used for field angle adjustment. First, use the Camera Alignment Assist feature to make the angles of view of the two cameras as similar as possible. Then fine-tune errors that cannot be physically adjusted in software. - Timing controller control
The timing controller LC880 is controlled in real time and performs synchronous control of the devices that make up the system, such as cameras and lasers. -
laser intensity monitor
In LIF, the luminance value of each pixel is often converted to density, temperature, etc., and shot-to-shot variations in laser intensity lead to measurement errors. KoncertoLIF can measure and record the laser intensity during image acquisition and compensate for measurement errors due to fluctuations in laser intensity between frames. - Displaying high-bit images
High-bit images such as 16-bit can be displayed in an easy-to-understand manner. - Analysis/display function
Equipped with various functions necessary for micro-shot analysis, such as real-time camera image display function, pseudo-color display, average calculation, calculation, correction of spatial luminance distribution variation, image correction, calibration (scale factor, scalar amount). - Confocal scanner control
Yokogawa Electric CSU series control is possible, and high speed and high spatial resolution of micro imaging are possible. - piezo focus scanner control
It is possible to scan the focal plane at high speed using a piezofocus scanner for microimaging. Scanning can be performed in conjunction with the frames of various cameras, and various program scans such as time-lapse shooting and micro PIV shooting are possible.
Principle of LIF
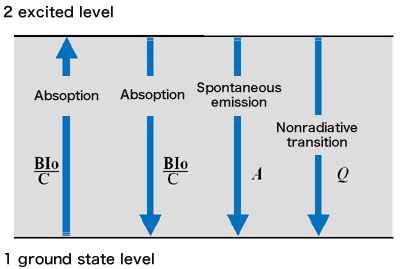
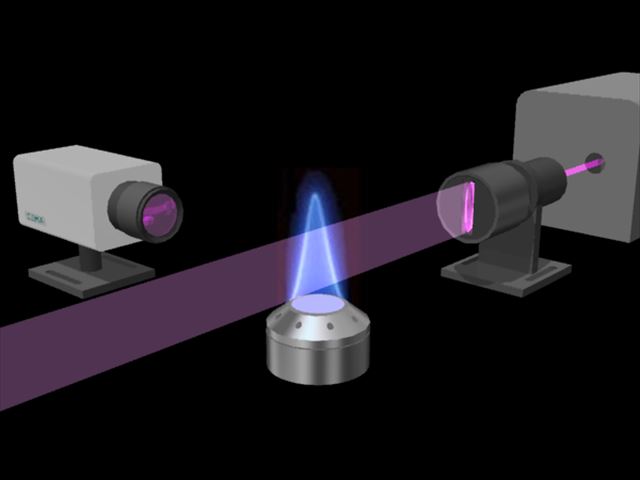
In general, LIF excites specific atoms/molecules with laser light, observes the luminescence caused by that, obtains the excitation spectrum, knows the concentration of atoms/molecules from the intensity, and knows the temperature from the spectrum distribution. It's a way. The excitation light is incident on the observation area where the target molecule exists, and the target atom or molecule is resonantly excited. The generated fluorescence is collected by a lens with a relatively large aperture installed perpendicular to the optical axis of the excitation light, and detected by a detector such as a camera. Based on the obtained image, measurement is performed by converting the brightness value of each pixel into density, temperature, etc.
application

- Radical measurement in engine combustion chamber
- Burner Radical Measurement
- Radical measurement in plasma
- Temperature measurement
- Concentration measurement
- pH measurement
- Mixed state measurement
LIF system components
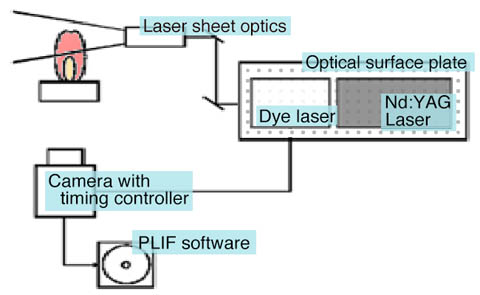
Illustrate the basic optical system for observing LIF. The excitation light is configured to be incident on the observation region where target molecules are present. A laser then resonantly excites the atoms and molecules of interest. Fluorescence generated by excitation is collected by a lens with a relatively large diameter set in the direction perpendicular to the optical axis of fluorescence generated by excitation, and detected by a detector such as a camera. Dye lasers capable of wavelength conversion over a wide range and their harmonics are used as excitation light sources.
System configuration example
■ Pulse laser
■ Dye laser
■ Control software (Koncerto-PLIF)
Timing controller
■ Laser intensity monitor function
■ Dedicated light sheet optical system
■ High sensitivity camera
■ Lenses, surface plates, workstations
List of LIF components
Various LIF specifications
Combustion measurements using PLIF can measure the molecular species of gases such as acetone, OH, CH, and NO, and their instantaneous distributions.
The system configuration/specifications differ depending on the gas type.
Please refer to the specification examples for each gas type.